On my most recent trip to the NIH, I was hoping maybe I would be disqualified for the TRAP-MS study, which evaluates four existing FDA-approved medications for their potential to reverse progression in multiple sclerosis. I don’t like that I have disease progression, and therefore qualify for the study. But if I am someone whose MS is progressing, I like the option of being first in line to take an FDA-approved medication that might help.
This was my second six month check-in to establish a baseline on my status with my new MS drug, Tecfidera.
Before I flew out, Dr. W, my NIH doctor, told me there was a chance I’d test out of the trial, since I didn’t have much progression to medicate. I wasn’t as optimistic about my lack of MS progression. My fall this January didn’t just fracture five bones in my face—it fractured all the routines I’d set up to live as healthily as I could with MS. I’d only just started going back to the gym. I hadn’t yet returned, wholeheartedly, to the Wahl’s-ish diet I’d been following.
During my summer exam, Dr. W had clucked at my balance— “your balance is shit”—and the lack of resistance in my right leg:—“so weak.” What would my balance be like after two months of not daring to challenge it? What would my strength be like after two months of barely any dog walks and zero visits to the gym? Thankfully, this setback didn’t prompt an MS relapse. But if there was going to be a time when my MS might be progressing, it would be now.
I’d been instructed to “take it easy” before my clinic visit. But that morning, I had pushed myself to the verge of immobility.

As I entered Building 10, my spirits rose. Maybe it was the profusion of plants, or maybe the profusion of people. I noted there was new art on the wall.

I noted there was still no additional entry in the display of Presidential Visits.

Luckily, the first appointment on my schedule was at Phlebotomy, which involved sitting, first in the waiting room, and then for the needle. That gave me some time to recover from all the walking I’d done through the airports, the Metro, and the NIH campus.
When my number was called, I got the same needle master as I’d drawn on my visit the previous summer. I recognized the fan letter he’d posted about his ability to make a two year old smile while drawing his blood.

The testimonial hangs strategically at eye level of the person waiting to get their blood drawn. It is terribly effective at arresting any impulse to cower or cringe. Nobody wants to come off as a bigger baby than a sick two year old.
Did I, like the sick two year old, leave smiling after having my blood drawn? I doubt it. I’ve not been known to smile until after I’ve had my second breakfast. I soon discovered that my favorite cafeteria—on the second floor in the new building—was closed for remodeling. I found my way to the alternate cafeteria in the basement of the old building. I loaded up on greens and root veggies and proteins. The meal provided all the rejuvenation I needed. By the time I stepped off the elevator on the fifth floor and rounded into the clinic, I no longer had a limp. Seeing Diane, a nurse I’ve known since my first NIH visit, put me in the mood to smile.
Since returning to real life after my big fall, I’ve become accustomed to being greeted with “Your face looks good!” If I chose to take “looks good” as meaning anything more than “not permanently damaged,” it is because I latch shamelessly onto the positive.
Diane obviously didn’t get the memo about the five facial fractures. Instead of saying, “Your face looks good!” she greeted me with a hug and a frank assessment. “We’re getting old!”
True enough! I, for one, don’t mind looking (or getting) old. I’ve earned my silver stripes. Besides, those strands provide an instant, socially acceptable explanation for a slow or unsteady gait. It’s not that I am MS closeted—it’s just that not every distressed person stuck behind me on the staircase really wants or needs an explanation of the ravages of autoimmune disease when an assumption about the ravages of age will do.
Diane didn’t look any older, and I told her so. Diane is remarkably stable. She just doesn’t change. Case in point: the day I met her, she had just won a prize in a weight-loss competition between nurses on her floor. Here’s the catch: Diane had won by losing a mere pound and a half. If Diane has had any weight fluctuations since then, they have probably been within the same range. Diane has had the same haircut as long as I’ve known her: same bangs, same color, same length. Diane stays Diane. I wouldn’t want her any other way.
Jen, the other dear nurse I’ve bonded with from the start—swooped in to agree with Diane’s assessment, “We are getting old!” as she grazed my cheek with a kiss. Jen’s hair was red that afternoon—her hair is a new color, a new style, every time I see her. Jen tends to pretend she’s disorganized or absentminded or late. True to form, she crafted an overly-elaborate explanation for why she couldn’t linger as she dashed off down the hall on her sturdy Doc Martens.
Diane wondered aloud, “How long have you been coming here, anyway?”
I guessed, “Maybe nine or ten years.”
Diane pulled up my file. “Since 2010. Nine years.”
When I’d first come to the NIH, I’d been chasing daclizumab, the only drug that had managed to stop my MS relapses. After the NIH had requisitioned all the supplies of it, I was more than grateful that there was room in the trial of the drug for me. For the three years of that trial, Diane and Jen had taken my weight and my blood every month. Once that trial was over, I was permitted to continue to take daclizumab though the NIH “safety study” while we all waited for the FDA approval. I flew in every six months for monitoring and new drug supplies. Once the drug was approved, we took what may yet be a final group photo.
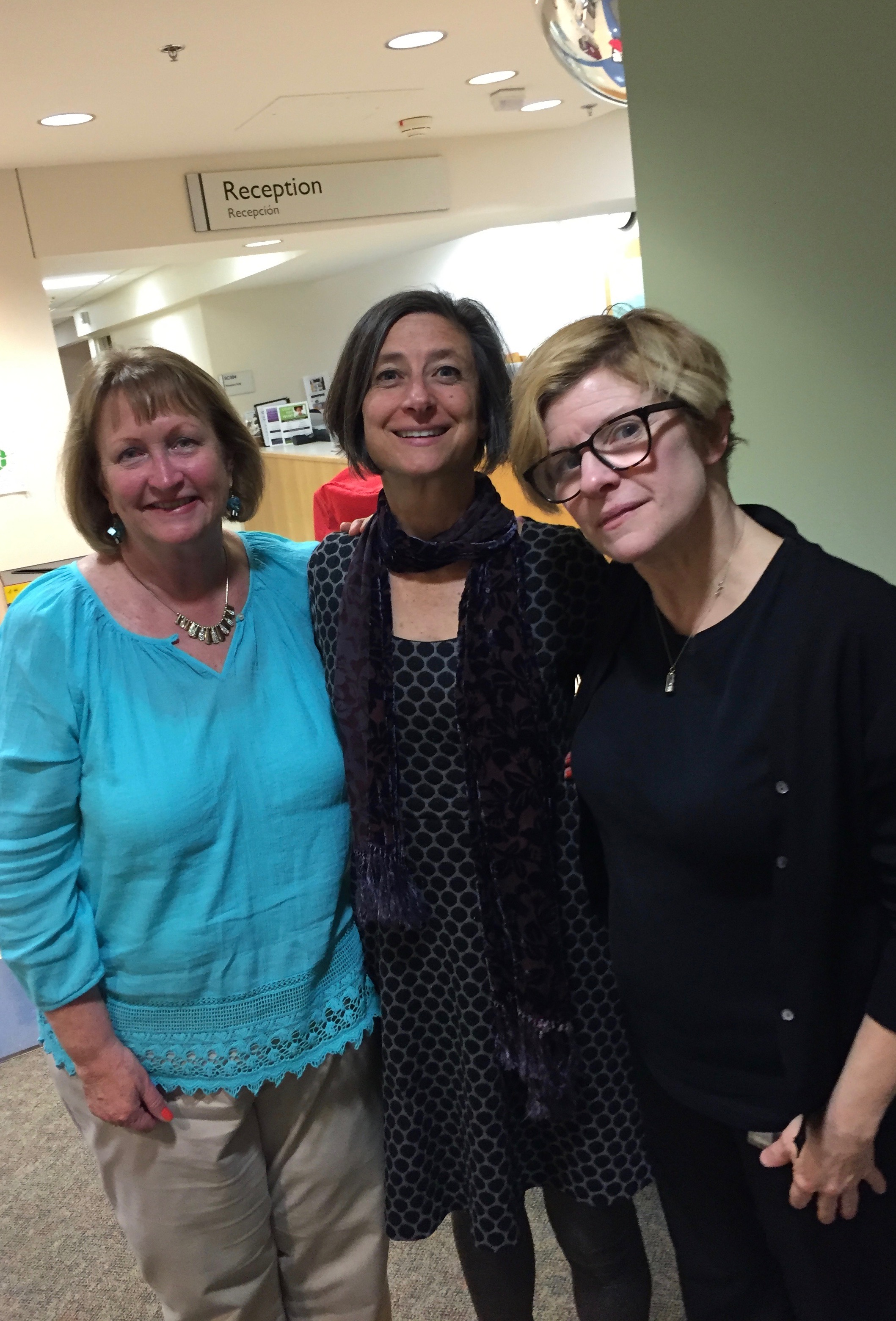
Thanks to TRAP-MS, we were back together again.
While Diane was at her computer, she got a notification on my labs. She scrolled down two screens of data. “Your labs look good.”
I thanked her, and told her how, the last time I’d visited, I gotten my lab results on my phone just as I was about to get on my departing flight. The cholesterol was marked in red, and looked way high. I was freaking out that the diet I was on was going to give me a heart attack. Doctor Google hadn’t been much help.
“Stay away from Doctor Google. You should call us when you have a question.”
Diane scrolled up, “Your good cholesterol is what’s really high.”
Then Linh, one of the graduate students, stuck her head in the office. She was ready to give me my tests.
We started with the timed 25 foot walk. Considering I had been limping just an hour or so earlier, I wasn’t optimistic about the outcome. Still, I’ve been conditioned to give these tests my all, so when it was time to march from one masking tape line to the other, I barreled along the hall like I was on my way to lift a screaming baby out of a vat of boiling water. Then I turned around and barreled right back. My fear of falling paled in comparison to my fear of failing. I did not fall.
I announced, “My healthy appearance is a flimsy veneer.” Like the NIH don’t know that. Test by test, I went all out, competing with my better-rested summer self.
As Dr. W. examined me, she seemed gentler than last time. She didn’t push as hard for the resistance tests. She didn’t chide me for being weak. When I messed up on the heel-to-toe test, she let me re-take it. Twice. Not that I did any better.
Overall, Dr. W was enthusiastic about my condition. She told me maybe the rest I’d had was doing me some good. Or maybe the Tecfidera.
When Dr. W. called a few days later with the results of my visit, she assured me the MRI looked stable. She told me I’d actually performed better on some tests, like the peg test, than I ever have. But overall, my numbers still nudged a bit in the direction of progression. As long as there is a progression of my disease, I will continue to qualify for the study,
Dr. W wished me luck in getting disqualified from the study when she sees me next time in six months. As much as I love this crew, I would love to be too healthy to see them.



 We made it to our destination early. I wouldn’t have to wait for the 10:30 van to the NIH. I could easily make the 8:30.
We made it to our destination early. I wouldn’t have to wait for the 10:30 van to the NIH. I could easily make the 8:30. I consulted my email from the travel office, which instructed me to wait on level three between doors 5 and 6—on the far end of the airport. As I hustled in that direction, a uniformed agent asked me for my destination. When I said, “The NIH,” he directed me to wait between doors 3 and 4. I hesitated. Which to believe? The travel office or the guy on the ground? We locked eyes for a second. I figured that if I picked the NIH instructions over his word, I couldn’t come to him for help later. I showed him the instructions on my screen. He muttered, “Then do as it says.”
I consulted my email from the travel office, which instructed me to wait on level three between doors 5 and 6—on the far end of the airport. As I hustled in that direction, a uniformed agent asked me for my destination. When I said, “The NIH,” he directed me to wait between doors 3 and 4. I hesitated. Which to believe? The travel office or the guy on the ground? We locked eyes for a second. I figured that if I picked the NIH instructions over his word, I couldn’t come to him for help later. I showed him the instructions on my screen. He muttered, “Then do as it says.”












